Posts Tagged ‘FDA’
Systematic review finds more clinical harm than benefits in Alzheimer’s “treatments” lecanemab, aducanumab, and donanemab
Study questions benefit of new Alzheimer’s drug (UGA Today): Last summer, the U.S. Food and Drug Administration fully approved the first drug shown to slow the progress of Alzheimer’s. But new research from the University of Georgia suggests that patients and caregivers may not experience any benefit from the drug in their daily lives. The drug, Leqembi,…
Read MoreBeyond the clinic: Can digital therapeutics (DTx) help boost mental health in the workforce at scale?
Hoping to Avoid Pear’s Fate, Behavioral Health-Focused DTx Companies Look to Employer Market (Behavioral Health Business): The digital therapeutics (DTx) industry is at a crossroads after one of the most prominent companies in the space, Pear Therapeutics, filed for bankruptcy earlier this year. The turmoil comes after DTx began catching on in the behavioral health…
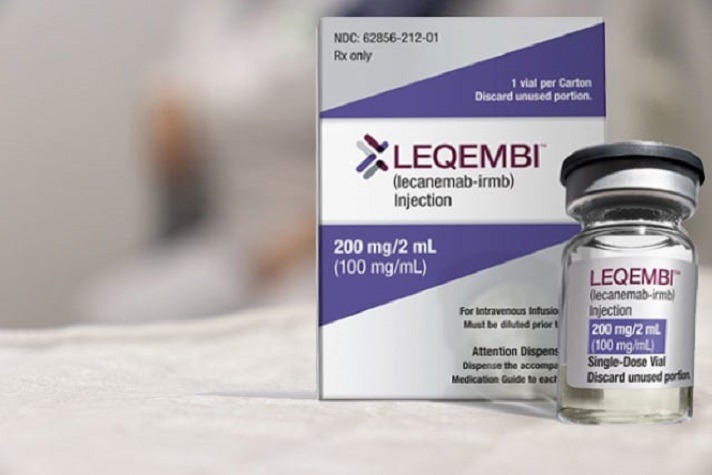
Read MoreHopes and Questions raised by Alzheimer’s drug Leqembi (lecanemab)
The FDA has approved Leqembi, the first disease-modifying treatment for early-stage Alzheimer’s and a precursor condition, mild cognitive impairment. Medicare has said it will pay for the therapy. Medical centers across the country are scrambling to finalize policies and procedures for providing the medication to patients, possibly by summer’s end or early autumn. It’s a…
Read MoreGrowing controversy over role of FDA and Medicare in promoting anti-amyloid drugs given limited benefit, high cost, severe side-effects
The War Over Whether Medicare Should Pay For The New Anti-Alzheimer’s Drugs (Forbes): The powerful Alzheimer’s Disease lobby is fighting a multi-billion-dollar battle on two fronts. It is quietly trying to limit restrictions the Food and Drug Administration puts on the use of new drugs aimed at slowing the progression of the brain disease. And…
Read MoreCMS: anti-amyloid drug Leqembi (lecanemab) doesn’t meet the “reasonable and necessary” standard required for wider Medicare coverage
CMS Sticks to Sharply Limited Coverage of New Alzheimer’s Drug, Leqembi (Managed Healthcare Executive): For now, CMS (Note: Centers for Medicare & Medicaid Services) is sticking to the coverage decision it made for Aduhelm (aducanumab) and applying it Leqembi (lecanemab). The decision limits Medicare coverage of the two Alzheimer disease’s drugs to Medicare beneficiaries who…
Read MoreOn cognitive-physical training, brain biomarkers, dementia, ketamine, brain teasers, riddles and more
Welcome to the last 2022 edition of SharpBrains e‑newsletter, featuring a few selected brain health news and fun teasers to challenge your brain and help us all think outside the box. #1. “Everyone agrees that Google Glass failed when it came to market, but to me, it’s a symbol of hope … It inspired people to…
Read More