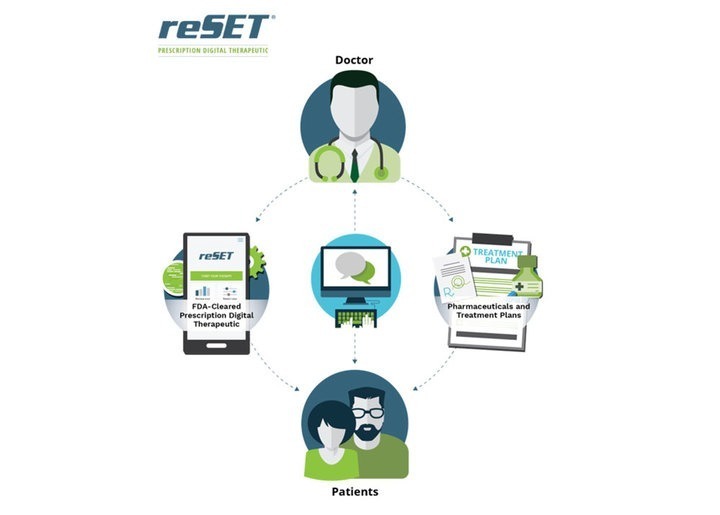
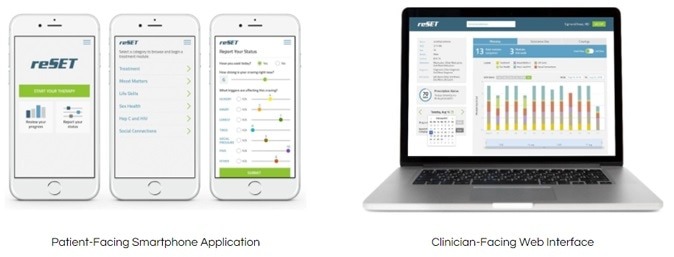
Posts Tagged ‘reSET-O’
Pear Therapeutics raises $175M and goes public via SPAC deal raising the profile of prescription digital therapeutics
Digital health firm Pear goes public, raising $175 million (pharmaforum): Pear will make its debut on the exchange today after combining with Thimble Point – a special purpose acquisition company (SPAC) – in a deal that sidestepped the conventional initial public offering (IPO) route and has propelled its valuation to around $1.6 billion … Pear specialises…
Read MorePrescription software firm Pear Therapeutics to go public via $1.6 billion SPAC deal, harnessing 3 FDA-authorized products and 14 candidates
Pear Therapeutics to Go Public in Roughly $1.6 Billion SPAC Deal (The Wall Street Journal): Medical technology company Pear Therapeutics Inc. has agreed to go public by merging with a blank-check company with ties to the Pritzker Vlock Family Office, betting on the growing role of prescription digital therapeutics. The proposed merger would give the…
Read MoreOhio joins Indiana and Kentucky in funding Pear Therapeutics’ digital addiction treatments
Ohio is third state to fund Pear’s digital therapeutics to tackle addiction (pharmaforum): The company has been pushing for more reimbursement of its three marketed DTx products in the US following a first FDA approval in 2017. Pear said that the Ohio Department of Mental Health and Addiction Services (OhioMHAS) and RecoveryOhio are providing access to…
Read MorePear Therapeutics raises $80M; finds cost savings of $2,150 per patient with opioid use disorder
Pear Therapeutics raises $80M in push for reimbursement (MedCity News): Pear Therapeutics raised $80 million in funding, which it plans to use as part of a push for coverage of its digital therapeutics. The Boston-based company has three FDA-cleared products, including digital therapeutics for substance use disorder, opioid use disorder, and most recently, insomnia.
Read MorePear Therapeutics raises $50M to develop and market a portfolio of digital therapeutics for mental health
___ Pear Therapeutics rakes in $50M to develop additional digital therapeutics (MobiHealthNews): “Boston- and San Francisco-based Pear Therapeutics, maker of a prescription digital therapeutic platform, announced today the closure of $50 million in Series B funding. The round was led by Singapore-based investment company Temasek, with participation from returning investors 5AM Ventures, Arboretum Ventures, and…
Read More