Deep learning model built on neuroimaging data identifies “Brain Age Gaps” as markers of Alzheimer’s disease (AD)
 Brain Age Gap is a Composite Biomarker for Dementia Pathology or Severity (GEN):
Brain Age Gap is a Composite Biomarker for Dementia Pathology or Severity (GEN):
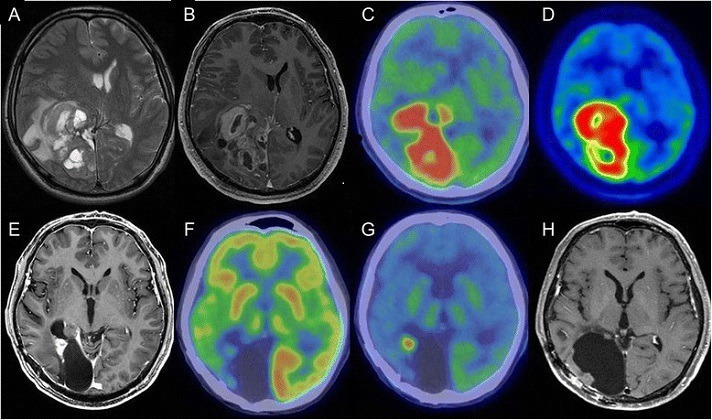
Mayo Clinic scientists have developed a computational model that predicts brain age using a large collection of neuroimaging data obtained using FDG-PET (fluorodeoxyglucose positron emission tomography) and structural MRI (magnetic resonance imaging). The deep learning-based model tests the relationship between brain age gaps in various forms of dementia, including mild cognitive impairment (MCI), Alzheimer’s disease (AD), frontotemporal dementia (FTD), and Lewy body dementia (LBD), as well as in normal brains.
… “The ability for deep learning to accurately predict age based on brain imaging data has been known for some time. However, looking at brain age gap or the difference between predicted and actual age, has been thought to have the potential to be utilized as a biomarker. Others have argued that such a brain age gap is only able to mark treatment-level biological differences and is unable to track changes in state and therefore should not be interpreted as accelerated brain aging,” (Senior author of the study, Dr. David) Jones said. “The main finding of our study is that we could indeed find evidence that high brain age gap is behaving as an accelerated brain aging biomarker.”
The Study:
Deep learning-based brain age prediction in normal aging and dementia (Nature Aging).
Abstract: Brain aging is accompanied by patterns of functional and structural change. Alzheimer’s disease (AD), a representative neurodegenerative disease, has been linked to accelerated brain aging. Here, we developed a deep learning-based brain age prediction model using a large collection of fluorodeoxyglucose positron emission tomography and structural magnetic resonance imaging and tested how the brain age gap relates to degenerative syndromes including mild cognitive impairment, AD, frontotemporal dementia and Lewy body dementia. Occlusion analysis, performed to facilitate the interpretation of the model, revealed that the model learns an age- and modality-specific pattern of brain aging. The elevated brain age gap was highly correlated with cognitive impairment and the AD biomarker. The higher gap also showed a longitudinal predictive nature across clinical categories, including cognitively unimpaired individuals who converted to a clinical stage. However, regions generating brain age gaps were different for each diagnostic group of which the AD continuum showed similar patterns to normal aging.


