Integrating music, movement and stroke rehabilitation, MedRhythms raises $25M to develop and commercialize digital therapeutic
 MedRhythms raises 25m to get patients back in tune after a stroke (TechCrunch):
MedRhythms raises 25m to get patients back in tune after a stroke (TechCrunch):
MedRhythms secured $25 million in Series B funding to advance its digital therapy platform aimed at measuring and improving someone’s ability to walk after they have experienced a neurologic injury or disease … Company co-founder and CEO Brian Harris was a neurologic music fellow at Spaulding Rehabilitation Hospital in Boston, treating people with stroke and brain deficits with music. He began getting questions from patients and families on how they could access similar care outside of the hospital. Not seeing a suitable alternative, he started MedRhythms with entrepreneur Owen McCarthy in 2016.
“There is no other stimulus that engages the brain like music does,” Harris said. “When someone is engaging in music, it aids in neuroplasticity to create new connections and strengthen old ones. Neuroplasticity is how we can learn new things or why people with brain deficits can improve.”
The Announcement:
MedRhythms Closes $25M Series B Funding Led by Morningside Ventures and Advantage Capital to Advance Digital Therapeutics Platform (press release):
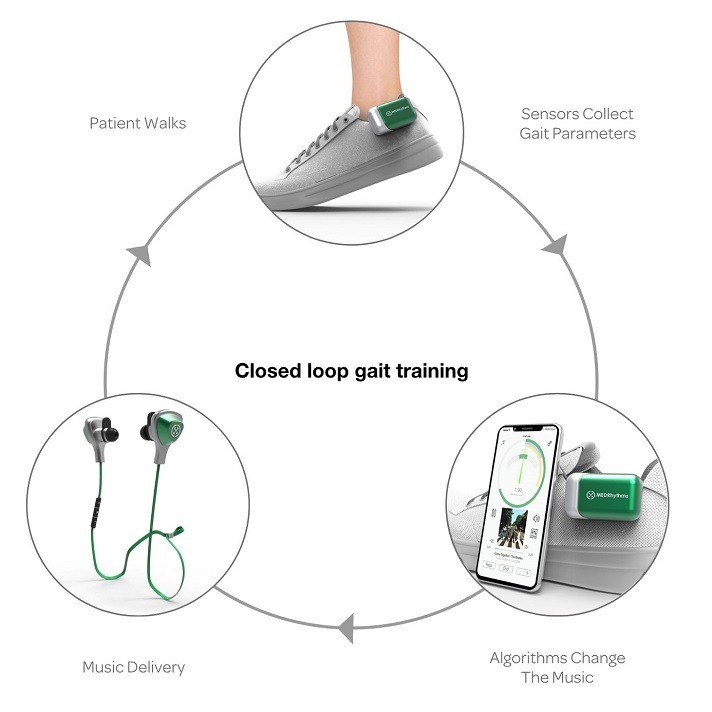
MedRhythms, a leader in developing prescription digital therapeutics that use sensors, software, and music to measure and improve walking for patients with a neurologic injury or disease, announced today the closing of a $25 million Series B financing round co-led by Morningside Ventures and Advantage Capital, with participation from existing investor Werth Family Investment Associates … MedRhythms’ digital therapeutic platform is designed to digitize an evidence-based intervention known as Rhythmic Auditory Stimulation, which uses music to target neural circuitry that controls movement. This mechanism is known as “entrainment,” a neurologic process in which the auditory and motor systems of the brain are coupled in synchrony with an external rhythmic cue. Entrainment can, over time, both enhance neuroplasticity and lead to improved functional outcomes in walking…
“MedRhythms has developed a powerful digital therapeutics platform founded on the premise that targeting electrophysiological networks in the brain can be broadly effective against neurologic disorders,” said Stephen Bruso of Morningside Ventures … MedRhythms has received Breakthrough Device designation from the U.S. Food and Drug Administration (FDA) for its patented digital therapeutic to treat chronic stroke walking deficits. This product is the first in the company’s pipeline of digital therapeutics for neurologic disease and injury. The impact of the device on chronic stroke walking impairments are currently being studied in a randomized controlled trial (RCT) at the nation’s top rehabilitation hospitals and research centers. The company also has also expanded its pipeline into neurological conditions such as Parkinson’s, acute stroke and multiple sclerosis, including a neuroimaging study with Massachusetts General Hospital.
News in Context:
- Ten neurotechnologies about to transform brain enhancement & health
- The National Academy of Medicine (NAM) shares discussion paper to help empower 8 billion minds
- AI-powered Woebot raises $90M to scale up digital self-therapy platform
- FDA clears MindMaze GO neurorehabilitation platform, easing access to continued outpatient therapy


